Romidepsin for Injection Available from Fresenius Kabi
First generic equivalent to ISTODAX® in the U.S. is latest addition to the most comprehensive generic injectable oncology portfolio in the industry
July 12, 2022
LAKE ZURICH, Ill., July 12, 2022 – Fresenius Kabi announced today the immediate availability in the United States of Romidepsin for Injection, the newest addition to the company’s broad portfolio of injectable oncology medicines.
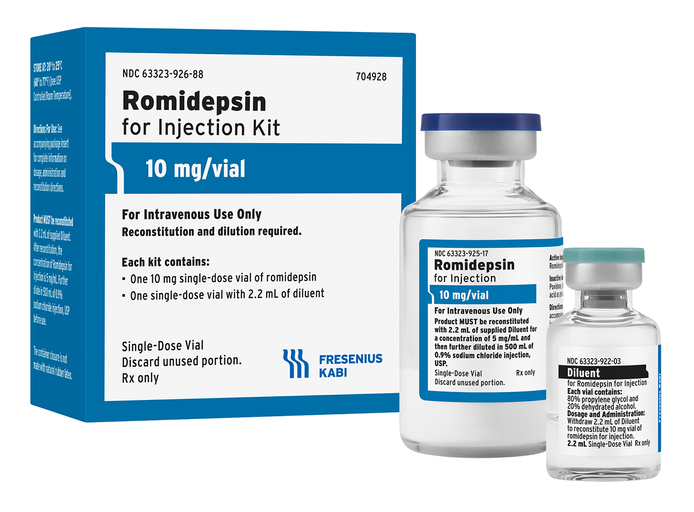
Fresenius Kabi Romidepsin for Injection is the first approved generic equivalent for ISTODAX®, providing clinicians and patients with a more affordable treatment option. Fresenius Kabi Romidepsin is supplied as a kit including one 10 mg single-dose vial of Romidepsin and one 2.2 mL vial of diluent.
“Generic oncology medicines continue to be vitally important to the protocols used in treating many forms of cancer,” said John Ducker, president and CEO of Fresenius Kabi USA. “As a leading U.S. developer and supplier of oncology injectable medicines, Fresenius Kabi remains committed to continuing to expand access to affordable oncology therapies.”
Fresenius Kabi Romidepsin is formulated, filled and finished in the United States. The company has invested nearly a billion dollars in U.S. manufacturing and distribution to further strengthen the domestic supply of essential medicines and technologies. To learn more, visit www.moreinamerica.com.
About Romidepsin for Injection
Romidepsin for Injection is a histone deacetylase (HDAC) inhibitor indicated for the treatment of cutaneous T-cell lymphoma (CTCL) in adult patients who have received at least one prior systemic therapy.
Important Safety Information
Myelosuppression: romidepsin can cause thrombocytopenia, leukopenia (neutropenia and lymphopenia), and anemia; monitor blood counts during treatment with romidepsin; interrupt and/or modify the dose as necessary.
Infections: Fatal and serious infections. Reactivation of DNA viruses (Epstein Barr and hepatitis B). Consider monitoring and prophylaxis in patients with evidence of prior hepatitis B.
Electrocardiographic (ECG) changes: Consider cardiovascular monitoring in patients with congenital long QT syndrome, a history of significant cardiovascular disease, and patients taking medicinal products that lead to significant QT prolongation. Ensure that potassium and magnesium are within the normal range before administration of romidepsin.
Tumor lysis syndrome: Patients with advanced stage disease and/or high tumor burden are at greater risk and should be closely monitored and appropriate precautions taken.
Embryo-fetal toxicity: Can cause fetal harm. Advise females of reproductive potential and males with female partners of reproductive potential of potential risk to a fetus and to use effective contraception.
The most common adverse reactions (≥ 30%), excluding laboratory abnormalities, are nausea, fatigue, infections, vomiting, anorexia, electrocardiogram ST-T wave changes, dysgeusia, constipation and pruritis. Grade 3-4 laboratory abnormalities (≥10%) include lymphopenia, neutropenia, anemia and thrombocytopenia.
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800‐551‐7176, option 5, or FDA at 1‐800‐FDA‐1088 or www.fda.gov/medwatch.
Carefully monitor prothrombin time (PT) and International Normalized Ratio (INR) in patients concurrently administered romidepsin and warfarin or coumarin derivatives.
Monitor for toxicities related to increased romidepsin exposure when co-administering romidepsin with strong CYP3A4 inhibitors.
Avoid use with rifampin and strong CYP3A4 inducers.
This Important Safety Information does not include all the information needed to use Romidepsin for Injection safely and effectively. Full prescribing information is also available at www.fresenius‐kabi.com/us.
About Fresenius Kabi
Fresenius Kabi (www.fresenius-kabi.com/us) is a global health care company that specializes in medicines and technologies for infusion, transfusion and clinical nutrition. The company’s products and services are used to help care for critically and chronically ill patients. The company’s U.S. headquarters is in Lake Zurich, Illinois. The company’s global headquarters is in Bad Homburg, Germany.
###
ISTODAX® is a registered trademark of Celgene.
