Biosimilars

Devoted to life-changing treatments
At Fresenius Kabi, our purpose is caring for life. And with our BioSpecialized™ approach to biosimilars, we are turning the promise of effective and affordable biologic therapies into reality for more patients.
We bring deep expertise in complex medicines, state-of-the-art supply chain and manufacturing capabilities, and above all, exceptional customer support to everything we do. That’s what makes us BioSpecialized.
We have already launched biosimilars in Europe, Australia and Canada. And, with an extensive portfolio of intravenous and injectable medicines, plus over 100 years of pharmaceutical experience and innovation, we are ideally positioned to deliver the benefits of biosimilars to you and your patients.
Click here for more information about BioSpecialized and our growing portfolio of biosimilars.
We’re Making High-Quality Biologics More Accessible
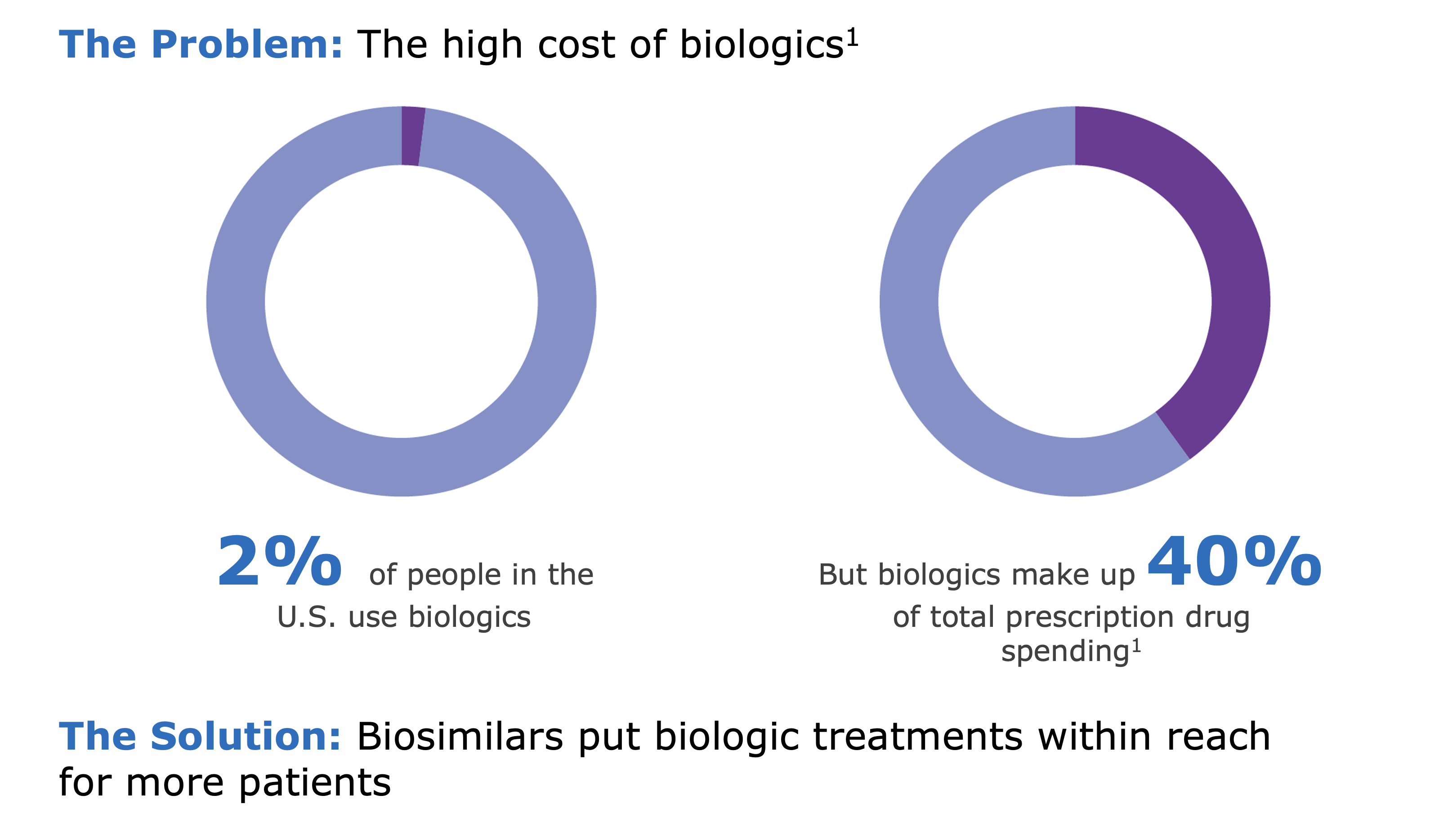
With demonstrated biosimilarity to the reference products at lower costs to patients and providers, biosimilars play an important role in the future of health care by improving access to medications and encouraging investments in research and innovation.
Biosimilars can potentially offer up to 30% price reductions when compared with reference products2
These savings allow physicians to follow established treatment guidelines without compromises based on cost, and encourage investment and innovation in other areas of medicine. Thanks to biosimilars, direct spending on biologic drugs in the U.S. is estimated to drop by $54 billion between 2017 and 2026.3
Above all, we are committed to making high-quality health care affordable and accessible to more patients coping with critical and chronic diseases.
Follow our BioSpecialized journey here.
References
- lnserro A. Enzi, Hassan introduce bipartisan bill to improve biosimilar education. American Journal of Managed Care (AJMC). Published May 27, 2019. Accessed May 20, 2022. https://www.centerforbiosimilars.com/view/enzi-hassan-introduce-bipartisan-bill-to-improve-biosimilar-education
- Kabir ER, Moreino SS, Siam MKS. The breakthrough of biosimilars: A twist In the narrative of biological therapy. Biomolecules. 2019;9{9):410. https://pubmed.ncbi.nlm.nih.gov/31450637/
- Building a better market for biologics: the US vs. Europe. Bionest blog, July 1, 2019. Accessed May 20. 2022. https://bionest.com/biologics-us-vs-europe
3242-BIOS-08-09/22
